Lactobacillus vs. Oscillibacter! Does (Saturated) Fat Tip The Scale Towards Leaky Gut, Obesity & Visceral Inflammation?
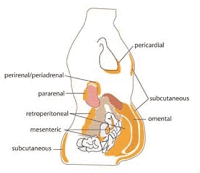 |
| Image 1: Location of the different fat depots of the human body (sorry Evilyn, had to borrow this from your Carbsane blog ;-) |
You cannot spot reduce fat, but can you "spot inflame" it?
In order to assess the effects of normal vs. high fat (60%) diets on gut Permeability and microbiota the Yan Y Lam and his (or her?) colleagues from the University of Sidney kept a group of 16-week old Female C57BL/6J mice (those are the "normal" lab mice used in these experiments) on a regular chow diet (control; 10% fat) or a high saturated fat (34%) high fat diet (HFD; 60% of total energy from fat). After 12 weeks, the rodents in the HFD group were obese and diabetic, had elevated insulin and amyloid A3 serum levels and reduced adiponectin.
What are amyloid A3 and adiponectin? Not to long ago, I would probably have had to explain what insulin is, but in these days of carbophobia, I better stick to the aliens, amyloid A3 and adiponectin. While the former is an acute phase protein elevations of which are indicative of adipocyte (=fat cell) inflammation and have also been observed in Creutzfeldt Jacob and Alzheimer patients (Baker. 2003), adiponectin is a negative control factor for body fat and energy expenditure, with increasing adiponectin expression from adipose tissue being linked to increases in fatty acid oxidation, improvements in glucose metabolism and reduced triglyceride storage within existing body fat stores.
While these are results we have seen in countless of studies before, the interesting part of the study began with the collection of stool samples in the last three days and the "termination" of the animals at the end of the 12-week study period. |
| Figure 1: Gut microbiota composition in control and high saturated fat diet (HFD) fed mice after 12-weeks (Lam. 2012). |
At the phylum level, HFD mice had more Firmicutes (73 6 1.5% vs 68 6 2.3% for controls; P = 0.041) and fewer Bacteroidetes (156 1.7% vs 196 1.3% for controls; P = 0.026) and thus a significantly higher Firmicutes: Bacteroidetes ratio ( P = 0.019). HFD also induced significant shifts in fecal microbiota composition at the genus level of taxonomic resolution. Within members of the Firmicutes, notably there was a 75% decrease in Lactobacillus ( P ,0.001) and a 279% increase in Oscillibacter( P = 0.004) as compared to controls and these changes in abundance were closely associated with weight gain.These gut microbiotal changes, in turn, were closely associated with differences in gut permeability, as measured according to Wang et al. 2001 (Wang. 2001), showing positive correlations (r = 0.52; P = 0.013) with the abundance of Lactobacillus and negative correlations with Oscillibacter (r = - 0.55;P = 0.007) in the proximal colon.
Notably, increased Oscillibacter abundance was also associated with a reduction in the mRNA expression of ZO-1 (r = - 0.37; P = 0.039) and, although not statistically significant, a similar trend was observed with proglucagon (r = - 0.34; P = 0.061).While the reduced expression of the macromolcule ZO-1 is symptomatic, maybe even causative for the "leakiness" of the gut (Assimakopoulus. 2011), the reduction in proglucagon, a pro-peptide to GLP-1 the fat-burning effects of which I have discussed in a previous blogpost (cf. "Eat more, burn more and lose fat like on crack"), could partly explain the diet induced hyperphagia (unsatiable hunger) and pronounced weight gain, which is yet perpetuated by what could eventually turn out to be a life-threatening down-stream effect of the leaky gut: Inflammation in the adjacent visceral adipose tissue:
 |
| Figure 2: Macrophage infiltration (per 100 adipocytes) and adipocyte area in the different fat depots of the mice on normal (control) and high fat diet (HFD) at the end of the 12-week study period (data adapted from Lam. 2012). |
Causal, corollary, chicken or egg?
 |
| Image 2: Until we know it's not the saturated fat that tips the scale in favor of Oscillibater you better have Sauerkraut with your Weißwurst ;-) |
So happy to be a "Kraut" ;-)
Whether it is a or even the central player and whether the saturated fat content and not the lack of other nutrients cause the undesirable shift in the microbiota composition of the gut still has to be determined. I guess, in the meantime you better make sure to have Bavarian Weißwurst with Sauerkraut ;-)
Update: If you want to know more about the correlation between different food-types and nutrients and the gut microbiom, check out my previous post on the "Gut Type Diet".


