 |
| Breakfast of a supplement junkie. |
In the latest issue of
Andrology, scientists from the
University of Tennesse e Graduate School of Medicine attempt to provide an overview of the most common designer anabolic steroids on the market. As Kim et al. point out, recent evidence suggests that these agents "may be the most common cause of hypogonadism in men of reproductive age" (Rahnema. 2015).
Despite all recent regulatory efforts the FDA is having a hard time to keep up with the tricks of the "supplement" industry who releases one structurally slightly modified, but effective AAS derivative after the other.
Learn more about natural muscle builders at the SuppVeristy

Optimizing Rest for Size and Strength Gains

Alternate Squats & BP for GAINS!

Farmer's Walk or Squat? Is Strong- men T. For You?

Full ROM ➯ Full Gains - Form Counts!

Battle the Rope to Get Ripped & Strong

Up Your Squat by 25% With Sodium Bicarbonate
More specifically, most designer steroids are orally formulated with varying degrees of bioavailability. Some compounds are 17-alpha alkyl ated to improve the oral bioavailability of injectable pharmaceutical androgens, but this modification increases the toxicity of the androgen (Kazlauskas. 2010).
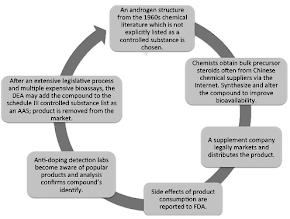 |
| Figure 1: Cyclic process of designer steroid discovery, synthesis, distribution, and regulation (Rahnema. 2015). |
"The designer steroid methasteron (Superol) is an example of the hepatotoxicity associated with the consumption of 17-alpha alkylated steroids. This oral AAS, also known as methyldrostanolone, was sold over-the counter until 2012 and is a methylated (17-alpha alkylated) version of the injectable steroid drostanolone (Masteron).
Consumption of methasteron as a dietary supplement for bodybuilding has been reported to cause severe hepatotoxicity, cholestasis, and acute renal failure" (Rahnema. 2015).
By slightly modifying the original chemical structure of potent androgens "supplement" companies have been successfully selling potent androgens without technically selling controlled substances by using pro-drugs that are unclassified compounds in the bottle, but in vivo are metabolized to Schedule III Controlled AAS for decades.
No, I am not trying to vilify these compounds. I don't want to point with a finger at people who are using them, either. I just want you to know that the evidence that the dreaded "Superdrol" is not the only agent that can harm your health is there - even though it may be scarce. As a responsible grown up person, it's obviously up to you whether you want to use a designer steroid or not. Just don't make the mistake and believe that popping pills alone could make up for your messed up diet and training regimen which hasn't yielded any results over the past two years.
Compounds like boldione or prostanozolol which have been banned in 2010 and 2012, respectively, are only the tip of the iceberg (Baume. 2006; Geyer. 2008; Kazlauskas. 2010). More recently other agents have been dominating the market. Agents like ...
- dimethazine - 2a,17a-dimethyl-5a-androstan-17b-ol-3,3 0-azine or 2a,17a-dimethyl-5a-androstan-17b-ol-3-one-azine aka DMZ,
- methylepitiostanol - 2a,3a-epithio-17a-methyl-5a-androstan-17b-ol aka Epistane,
- methoxygonadiene - 13b-ethyl-3-methoxy-gona-2,5(10)-dien-17-one aka Methadrol,
- methylclostebol - 4-chloro-17a-methyl-androst-4-en-17b-ol-3-one aka P-Mag,
- methylstenbolone - 2,17a-dimethyl-17b-hydroxy-5a-androst-1-en-3-one aka M-STEN,
- mentabolan - 7a-methyl-estra-4-en-3,17-dione ake MENT, and
- trestione - 7a-methyl-19-norandrostenedione aka TREST
have been found in over-the-counter dietary supplements that have been and in many cases still are legally sold in the United States and oversees.
 |
| Table 1: Currently unscheduled designer steroid | a designer steroids from the WADA Prohibited List (Rahnema. 2015) |
As the scarce data from scientific studies in
Table 1 suggests all of them may have severe side effects which include hepatotoxicity (=liver damage), cholestasis (=a condition where bile cannot flow from the liver to the duodenum), renal failure, hypogonadism (=low testosterone), gynecomastia (
learn more), and infertility.
 |
| Table 2: Parent compounds, testosterone, nandrolone and associated compounds from Baume et al. (2006). |
As Kim et al. point out, some of these side effects may be reversible, more aggressive use, though, may result in more permanent end-organ damage as has been previously described for the case of aggressive AAS users (Rahnema. 2015). Specifically, the impact of AAS use on the reproductive and hormonal health of men is, according to the authors of this review, "underestimated in the literature owing to previous studies’ failure to account for designer steroid use" (Rahnema. 2015).
 |
| It's actually quite funny that many of the AAS users on "the boards" appear to be more afraid of developing gyno than other significantly more severe health issues | learn more about gyno. |
Overall, we still know very little about "designer steroids". What we do know, though, is that they all hold the potential to mess up your liver and hormonal health big time.
In view of the fact that previous studies show that about 30% of classic AAS users will develop a dependence on their drugs of choice (Kanayama. 2001 & 2009), it is also questionable whether first time users will stick to probably "safer" short time cycles. Rather than that, a brief glimpse at the relevant bulletin boards reveals that most first time users will not just run additional cycles, but begin stacking one AAS with the other. A practice that entails which may lead to liver and heart damage, as well as ischemic stroke .. to mention only the most severe side effects of uncontrolled, excessive steroid abuse |
Comment on Facebook!
References:
- Baume, Norbert, et al. "Research of stimulants and anabolic steroids in dietary supplements." Scandinavian journal of medicine & science in sports 16.1 (2006): 41-48.
- Buzby, G. C., C. R. Walk, and H. Smith. "Totally synthetic steroid hormones. X. 1 Some (plus or minus)-13beta-ethyl-7alpha-methylgonane derivatives." Journal of Medicinal Chemistry 9 (1996): 782-784.
- Campbell JA, Lyster SC, Duncan GW & Babcock JC. "7a-Methyl-18-norsteroids: a new class of potent anabolic and androgenic hormones." Steroids 1 (1963): 317–324.
- Cavalcanti de Albuquerque , Gustavo, et al. "Detection of designer steroid methylstenbolone in “nutritional supplement” using gas chromatography and tandem mass spectrometry: elucidation of its urinary metabolites." Steroids 78.2 (2013): 228-233.
- De Ruggieri P., et al. "A new steroid having proteo-anabolic action: dimethazine." Archivio di scienze biologiche 47 (1963): 1.
- Edgren, R. A., et al. "Biological effects of synthetic gonanes." Recent progress in hormone research 22 (1965): 305-349.
- Geldof L, Lootens L, Polet M, Eichner D, Campbell T, Nair V, Botre F, Meuleman P, Leroux-Roels G, Deventer K & Eenoo PV. "Metabolism of methylstenbolone studied with human liver microsomes and the uPA(+/+) -SCID chimeric mouse model." Biomed Chromatogr 28 (2014): 974–985
- Geyer, Hans, et al. "Nutritional supplements cross‐contaminated and faked with doping substances." Journal of Mass Spectrometry 43.7 (2008): 892-902.
- Lootens, Leen, et al. "Metabolic studies with promagnon, methylclostebol and methasterone in the uPA+/+-SCID chimeric mice." The Journal of steroid biochemistry and molecular biology 127.3 (2011): 374-381.
- Mascher, R., C. Lupo, and P. De Ruggeiri. "Biological activity of dimethazine in the protein anabolic sense." Bollettino della Società italiana di biologia sperimentale 38 (1962): 988.
- Okano M, Sato M, Ikekita A & Kageyama S. "Analysis of non-ketoic steroids 17alpha-methylepithiostanol and desoxymethyl- testosterone in dietary supplements." Drug Test Anal 1 (2009): 518–525.
- Kazlauskas R. "Designer steroids." Handb Exp Pharmacol 195 (2010): 155–185
- Kurachi, Keiichi, et al. "Effects of 2alpha, 3alpha-epithio-5alpha-androstan-17beta-ol (epitiostanol) on hypothalamo-pituitary-gonadal axis in humans." Acta obstetrica et gynaecologica Japonica 22.1 (1975): 42-48.
- Rahnema et al. "Designer steroids – over-the-counter supplements and their androgenic component: review of an increasing problem." Andology (2015): Accepted Article.
- Segaloff A. "The enhanced local androgenic activity of 19-nor steroids and stabilization of their structure by 7a- and 17a-methyl substituents to highly potent androgens by any route of administration." Steroids 1 (1963): 299–315.
- Ueda H, Hagino Y, Ono M & Kuwano M. "Human mammary cancer cell mutants with altered hormone receptor activity." J Biochem 100 (1986): 341–348.













