Cooking Fatty Fish & Omega-3s: Do Baking, Broiling, Frying in Different Oils, or Microwaving Affect DHA, EPA & Co?
 |
| The good news is: Frying fish is not as bad as you may have thought it was. The oxylipin content even decreases. |
You can learn more about fish & co. at the SuppVersity
That's also because the scientists from the University of Minnesota and the USDA's very own Human Nutrition Research Center addressed a hitherto only cursorily treated aspect of preparing high-omega-3 foods: the formation of oxylipins.
Oxylipins are oxidized products of PUFA (Gabbs et al., 2015) that can be used as a measurement of lipid oxidation, and indicate which fatty acids are most impacted by heat.
In that, it is important to note that the evidence Gabbs et al. collected for their 2015 paper in Advances in Nutrition clearly indicates that not all oxylipins are created equal - or as Flaskerud et al. summarize the results from Gabbs et al. and related studies: "Omega-6 (n-6)-derived oxylipins tend to have increased inflammatory and vasoconstrictive effects, while n-3-derived oxylipins tend to be anti-inflammatory" (Flaskerud 2017).
Cooking fish increases the bioavailability of organic pollutants: The effect of cooking on the fatty acid composition of fatty fish is, at least that's what the study at hand suggests, probably overestimated by many, a recent study that raises concern over the bioavailability of organic pollutants. In fact, the authors conclude that "the human health risk assessment based on bioaccessible concentrations of DDTs and PBDEs in fish showed that the maximum allowable daily fish consumption rates decreased from 25, 59, and 86 g day–1 to 22, 53, and 77 g day–1 for children, youths, and adults, respectively, after fish were cooked with oil" (Mi 2017). Whether oils other than the corn oil Mi et al. used in this first study to assess the effect of cooking on organic contaminant bioavailability would have the same effect warrants future research.
It is thus all the more important to examine in detail how the method of preparation affects the oxylipin content of fish fillets to determine if there is a superior method of preparation that maintains the beneficial lipids found in fish. For rainbow trout, which contains a comparatively high amount of omega-3s (1.6%, which is in the range of mackerel (1.8%), herring (1.5%), sardines (1.4%), albacore tuna (1.3%), and salmon (1.1%)), this effect has been studied before, but previous studies did not investigate pan-frying with various oils, microwaving, baking, and broiling - Which is why, Flaskerud et al. did exactly that:
Pan frying -- Eleven grams of test oil [peanut oil (PO), high oleic sunflower oil (HOSO), corn oil (CO), or canola oil (CaO)] were used to fry the fish fillet (50g) at 177°C (350°F).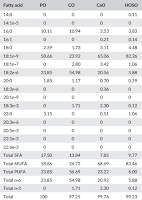
Table 1: Analyzed fatty acid content of oils used in pan-frying (Flaskerud 2017) - Why's that important? Because it adds to the fats and oxylipins from the fillets. - Baking -- In a preheated oven [177°C (350°F)] the fillet (50g) was placed in a 5.5″ × 4.5″ (14 cm × 11.4 cm) bake and serve container and cooked.
- Microwaving -- The 50 g fillet was placed in a 5.5″ × 4.5″ (14 cm × 11.4 cm) bake and serve container (Pactiv Pressware, Columbus, OH). Wax paper was used to cover the container. The sample was then placed into a 1,200-W high-power microwave and cooked in short intervals.
- Oven broiling -- In a preheated oven [260°C (500°F)] the fillet (50g) was placed in a Pyrex glass container which had 3 g of CO spread on the bottom.
For each cooking method, samples were cooked in triplicate until the core temperature reached 63°C (145°F) for 15 s. As soon as the desired temperature was attained by all cooking methods, the samples were cooled and frozen at −80°C until analyzed.
"Fatty acids and oxidized lipids were extracted from these samples and their respective raw fillet samples. Fatty acid content was determined using gas chromatography and oxylipin content by mass spectroscopy. The values obtained from each cooking method were compared to those obtained from the respective raw fillets using paired t tests" (Flaskerud 2017).The results show that it was well-worth to investigate both: the effect of the cooking method and its interaction with the cooking oils.
 |
| Figure 1: Relative changes in fatty acid composition (raw vs. cooked) in pan-fried fish fillets (Flaskerud 2017). |
- there were no changes in fatty acid composition when corn oil (CO) or canola oil (CaO) was used as a frying oil, in contrast, ...
- peanut oil (PO) reduced the concentration of α-linolenic acid (ALA | 18:3n-3), eicosadienoic acid (EPA | 20:2n-6), and dihomo-γ-linolenic acid (DGLA| 20:3n-6), and...
- high oleic acid sunflower oil (HOSO) reduced 18:3n-3, eicosapentaenoic acid (EAP | 20:5n-3), docosapentaenoic acid (DPA | 22:5n-3), docosahexaenoic acid (DHA | 22:6n-3), linoleic acid (LA | 18:2n-6), 18:3n-6, 20:2n-6, 20:3n-6, docosatrienoic acid (22:2n-6), and adrenic acid (22:4n-6) ...
If we assume that n-6 oxylipins are "bad" while n-3 oxylipins are "good", the study at hand suggests that the overall effect of cooking (at least the one that's mediated via the oxygenated natural products which are formed from n-6 and n-3 fatty acids) is positive (=health promoting).
This conclusion may not be obvious, though, if you look at the actual values of oxylipins in Figure 2. Firstly, you may easily confuse the raw (solid black) with the cooked (solid gray) bars, because we are so used to think of cooking as something that damages the cooked goods that our brain simply assumes the higher bars were the bars for the cooked fish.
Due to the correspondingly large standard deviations, the changes in the lipoic acid (n-6) oxylipin concentrations are the only statistically significant and practically relevant changes; so that overall, the effect of all cooking methods (n-6 oxylipins ↓; n-3 oxylipins ↔ )appears to be, as previously pointed out, identically (within statistically margins of error) beneficial.
 |
| Figure 3: Relative changes in the N-3 and N-6 PUFA concentration during frying with different oils (calculated based on Flaskerud 2017). |
The scientists thus pass the buck to high oleic acid sunflower oil (HOSO). Rightly so?
If we take a closer look at the data, it turns out that HOSO, like frying per se, reduced the total n-3 content, but unlike corn oil, for example, it improved the n-3/n-6 ratio of the fish fillets.
It is thus questionable, whether the scientists are right to single HOSO out. After all, the notoriously low n-3/n-6 ratio of our diet is at least as much of a problem as its generally low content of omega-3s (Simopoulos 2008); in fact, if you look at the corresponding data in Figure 3, you would have to conclude that the "best" way of frying - at least if "best" is defined as "having the most beneficial effect on the n-3/n-6 PUFA ratio" is frying in high-oleic acid sunflower (HOSO) or canola oil (you may remember HOSO and high MUFA canola oil as suggestions from my article about cooking oils).
Most important message: Cooked fish is a good source of omega-3s.
If we ignore the question "What's the best cooking oil to fry fish?" for the time being, the more important message of the study at hand is that cooking fish that's high in oxidatively not exactly stable n-3 fatty acids is less of a problem than many of you probably thought it was. Cooked fish can thus very well be a major source of (unoxidized) n-3s in our otherwise n-3-deficient diet.
What you must not forget, though, is that this doesn't negate the existing link between fried fish intake and type II diabetes I've addressed in March 2017. What it does, however, is to shrink the list of potential mechanisms which would now be reduced to AGEs (esp. for breaded fish fillets) and various mutagenic compounds (esp. heterocyclic amines of which it is important to know that their formation can be reduced by using antioxidant spices like rosemary, thyme, turmeric, garlic, ginger, or black cumin | Ahn 2005; Srinivasan 2017). Both can be formed during the frying process of fish and other foods and have not been tested in the study at hand. If you want to eat fish daily, it may thus be smart if you do what I have previously recommended and steam your fish - so far, this appears to be the safest way of preparing fish | Comment!.
- Ahn, Juhee, and Ingolf U. Grün. "Heterocyclic amines: 2. Inhibitory effects of natural extracts on the formation of polar and nonpolar heterocyclic amines in cooked beef." Journal of food science 70.4 (2005).
- Flaskerud, K, et al. "Effects of cooking techniques on fatty acid and oxylipin content of farmed rainbow trout (Oncorhynchus mykiss)." Food and Science Nutrition (2017).
- Gabbs, Melissa, et al. "Advances in our understanding of oxylipins derived from dietary PUFAs." Advances in Nutrition: An International Review Journal 6.5 (2015): 513-540.
- Mi, Xiu-Bo, et al. "Significance of Cooking Oil to Bioaccessibility of Dichlorodiphenyltrichloroethanes (DDTs) and Polybrominated Diphenyl Ethers (PBDEs) in Raw and Cooked Fish: Implications for Human Health Risk." Journal of Agricultural and Food Chemistry 65.16 (2017): 3268-3275.
- Simopoulos, Artemis P. "The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases." Experimental biology and medicine 233.6 (2008): 674-688.
- Srinivasan, Krishnapura. "Antimutagenic and cancer preventive potential of culinary spices and their bioactive compounds." PharmaNutrition (2017).









